  |
  |
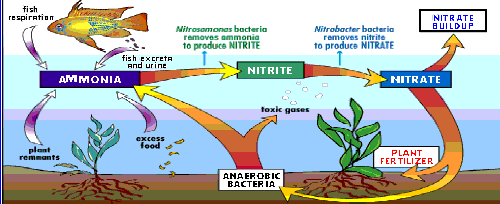
The Nitrogen Cycle is the most important process at work within your aquarium. This is nature's waste management system, removing organic toxins generated by your fish. The most lethal compound (unless you're a puffer) produced in the aquarium is ammonia (NH3). While fish release this compound in respiration, they release more of it in the form of solid and liquid waste. Other waste

Nitrosomonas
Nature has
produced (evolved) beneficial bacteria which feed on nitrogen, especially
ammonia. Nitrosomonas bacteria are found throughout almost every
ecosystem, both terrestrial and aquatic. They are largely lithotrophic
(found in soil) and aerobic. This bacteria forms colonies wherever there
is an adequate amount of ammonia and oxygen. This is one of the two
main constituents of your biological filter.
Nitrobacter
Nitrite
is the by-product produced by Nitrosomonas as it removes the ammonia from
your aquarium. Nitrite is also toxic to fish. Nitrobacter feed
on the nitrite produced by nitrosomonas, so it's not unusual to find the
two together. Although nitrite is toxic, your fish can withstand
roughly twice the amount of nitrite in their water when compared to ammonia.
The nitrite consumed by nitrobacter in turned into nitrate.
You can probably
see a cycle already starting as your fish are producing ammonia, which
is being eaten by nitrosomonas and turned into nitrite. The nitrobacter
in turn are feeding on the nitrite.
New aquariums require something called cycling. Cycling, as you can probably guess, is simply establishing the biological filter by introducing the nitrogen cycle to your aquarium.
Cycling your aquarium takes some time, and as seasoned fish keepers know, pays off in the long run. This is an excerpt detailing the cycling of an aquarium. (Pond, 1995)
Day 1 - 10
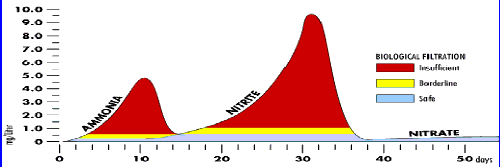
Note
the first 10 days on the above graph. Ammonia begin a rapid rise up to
lethal levels and then drops dramatically to close to a zero level. The
replication of the bacteria determine this rise and fall. Since they replicate
geometrically, the hypothetical first "bug" becomes 2 in 8 hours, then
4 (16 hours) then 16 (24 hours) the 32 (32 hours) then 64 (40 hours) and
so on..... It takes about ten days in relatively ideal conditions for the
bacterial to replicate to population where all the ammonia produced within
the aquarium is immediately reduced to nitrite. What is shown on the graph
is the sudden drop of ammonia concentration slightly after the 10 day maximum.
Day 10 to
21

The
next 21 days, after the ammonia spike drops to minimal, now show a low
to zero level of ammonia residue, but a steadily increasing concentration
of nitrite, rising much higher in concentration than the ammonia graph,
about double in fact. Nitrite is toxic, but not as toxic as ammonia, thus
the simple fact that the concentration can rise so high without a total
loss of fish. As nitrite becomes more evident, so do the populations of
nitrobacter removing the nitrite from the system and changing it into nitrate
[and energy for the bacteria]. As the populations grow, they gradually
become able to reduce nitrite as soon as it is created by nitrosomonas
acting on ammonia production. After 21 days from the beginning of nitrite
build-ups the spike falls rapidly to the low levels of the graph. From
there the nitrate levels start to rise over time.
There's a
Glitch

There is
a problem with the above simplistic view, Mother Nature has thrown a curve
into the
straightforward
Nitrogen Cycle reviewed above. The bacteria that reduces nitrite to nitrate,
nitrobacter
is inhibited by a free concentration of ammonia in the water. This is the
reason that the
nitrobacter
population is essentially kept at a zero level until day ten when the ammonia
spike reaches
the minimum
level. Once the ammonia inhibition is removed, then (and only then) nitrobacter
can begin to replicate. They are also lithotrophic so they require the
same things that nitrosomonas require, oxygen, their food source and clean
hard places to attach and populate.
After Day
31

Once the
nitrite is removed as fast as it is produced by nitrosomonas, the final
by-product of the
Nitrogen
Cycle is nitrate. It is a compound which is not easily reduced any further
by aerobic bacteria.
Because
of this, the nitrate levels begin to slowly rise and continue to build
over the rest of the life of
the aquarium.
The best way to get rid of nitrate is simply to practice proper water maintenance
procedures.
With regular water changes, nitrate is diluted, removing water with high
nitrate
concentrations
and replacing it with low nitrate conditioned tap water is one of the most
effective
ongoing
ways to eliminate nitrate. A second method tried in various ways is to
promote anaerobic
bacterial
growth, this takes nitrate and reduces it to nitrogen and other by-products.
In my opinion, this is dangerous, and extremely difficult to control. If
it doesn't go correctly, hydrogen sulfide and other toxic gasses are released
into the water column in bubbles. I have found proper water maintenance
is much better than relying on questionable anaerobic techniques to remove
nitrate.
Biological
Inoculation
When biological
inoculation products such as Hagen's Cycle are created, they must take
into account
this little
glitch. It precludes a "magic potion" where you simply add a culture of
bacteria and suddenly
the Nitrogen
Cycle's run-in period is eliminated. Cycle does noticeably reduce the run-in
time of a new
aquarium,
and it dramatically lessens the ammonia and nitrite spikes within the run-in
period, but it must be regularly added to the aquarium to overcome the
fact that ammonia inhibits nitrobacter. Using a regular weekly dose, the
little glitch mentioned earlier is overcome by the regular addition of
high
numbers
of both nitrosomonas and nitrobacter.